How Many Cc Are In A Mg
Treneri
Apr 05, 2025 · 4 min read
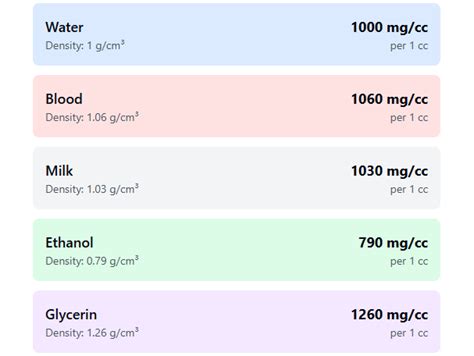
Table of Contents
How Many CCs Are in a MG? Understanding Cubic Centimeters and Milligrams
The question "how many cc's are in a mg?" is fundamentally flawed because cc (cubic centimeters) and mg (milligrams) measure entirely different physical quantities. It's like asking how many apples are in an orange – they simply aren't comparable. Let's clarify the distinction and explore the related concepts in detail.
Understanding Cubic Centimeters (cc)
A cubic centimeter (cc), also denoted as cm³ or mL (milliliter), is a unit of volume. It represents the volume of a cube with sides measuring one centimeter each. Volume measures the amount of three-dimensional space occupied by an object or substance. We use cc to measure the capacity of containers, the size of engines, and the volume of liquids and solids.
- Examples of cc usage:
- The engine displacement of a car (e.g., a 2000cc engine).
- The volume of a medicine bottle (e.g., a 100cc bottle).
- The volume of a container (e.g., a 500cc beaker).
Understanding Milligrams (mg)
A milligram (mg) is a unit of mass (or weight). It's one-thousandth of a gram (1 gram = 1000 mg). Mass measures the amount of matter in an object. We use mg to measure the mass of small objects, the dosage of medicine, and the weight of ingredients in recipes.
- Examples of mg usage:
- The dosage of a medication (e.g., a 500mg tablet).
- The weight of a small gemstone (e.g., a 20mg diamond).
- The weight of a spice in a recipe (e.g., 5mg of saffron).
Why You Can't Directly Convert cc to mg
The crucial difference lies in the nature of the units. You cannot directly convert between volume (cc) and mass (mg) without additional information. To relate volume and mass, you need to know the density of the substance.
Density is defined as the mass per unit volume of a substance. The formula is:
Density = Mass / Volume
Or, rearranging the formula to solve for mass:
Mass = Density x Volume
Therefore, to find the mass (in mg) of a substance given its volume (in cc), you need to know its density. The density is usually expressed in g/cm³ or kg/m³. Let's look at some examples:
Examples of Converting Volume to Mass Using Density
Example 1: Water
The density of water is approximately 1 g/cm³ (or 1000 mg/cm³). Let's say we have 10 cc of water. To find the mass:
Mass = Density x Volume = (1000 mg/cm³) x (10 cm³) = 10,000 mg
So, 10 cc of water has a mass of 10,000 mg (or 10 grams).
Example 2: Mercury
Mercury is much denser than water. Its density is approximately 13.6 g/cm³. If we have 5 cc of mercury:
Mass = Density x Volume = (13.6 g/cm³) x (5 cm³) = 68 g = 68,000 mg
Therefore, 5 cc of mercury has a mass of 68,000 mg (or 68 grams).
Example 3: A Less Dense Substance (e.g., Air)
The density of air is significantly lower than that of water or mercury, around 1.225 kg/m³ at sea level. To calculate the mass in mg, we need to convert the units:
1 kg = 1,000,000 mg 1 m³ = 1,000,000 cm³
Let’s say we have 1000 cc of air. Converting to cubic meters:
1000 cm³ = 0.001 m³
Now we calculate the mass:
Mass = Density x Volume = (1.225 kg/m³) x (0.001 m³) = 0.001225 kg = 1225 mg
Therefore, 1000 cc of air has a mass of approximately 1225 mg.
Practical Applications and Considerations
Understanding the relationship between volume and mass is vital in numerous fields:
- Medicine: Calculating drug dosages accurately requires knowing the density of the medication.
- Engineering: Designing engines, pipelines, and other systems needs precise calculations of volume and mass to ensure proper functionality and safety.
- Chemistry: Chemical reactions often depend on the mass and concentration of reactants, and precise volume measurements are crucial for accurate results.
- Food Science: Formulating food products requires careful consideration of both volume and mass of ingredients.
Common Mistakes and Misconceptions
It is crucial to avoid the common error of directly equating cc and mg. Remember that cc measures volume, while mg measures mass. Without knowing the density of the substance in question, any attempt to directly convert between these units will be incorrect.
Always be clear about the units you are using and ensure that you have the necessary information (density) to perform valid conversions between volume and mass.
Conclusion
The question "how many cc's are in a mg?" highlights a crucial concept in science and measurement: the difference between volume and mass. These two properties are distinct, and converting between them necessitates knowledge of the substance's density. By understanding the concepts of volume, mass, and density, you can accurately perform calculations and avoid common errors. Remember to always specify the substance to determine its density for accurate conversions between volume and mass. Accurate conversions are essential for diverse fields requiring precise measurements.
Latest Posts
Latest Posts
-
30 Kilos En Libras Cuanto Es
Apr 06, 2025
-
How Many Feet In A Linear Mile
Apr 06, 2025
-
How To Calculate Win Percentage With Ties
Apr 06, 2025
-
How Many Cups Is 3 4 Ounces
Apr 06, 2025
-
How Thick Is Fish Tank Glass
Apr 06, 2025
Related Post
Thank you for visiting our website which covers about How Many Cc Are In A Mg . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
