How Much Helium To Lift 1 Pound
Treneri
Apr 04, 2025 · 5 min read
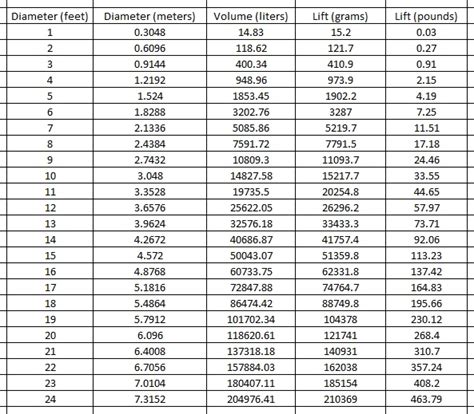
Table of Contents
How Much Helium to Lift 1 Pound? A Comprehensive Guide
Lifting objects with helium balloons is a captivating concept, often seen in celebrations and even scientific demonstrations. But the seemingly simple question, "How much helium to lift 1 pound?" requires a deeper dive into the physics and practical considerations involved. This comprehensive guide will explore the science behind helium buoyancy, the factors influencing lift capacity, and how to accurately calculate the required helium quantity for various lifting tasks.
Understanding Buoyancy and Helium's Lifting Power
At the heart of helium's lifting ability lies the principle of buoyancy. Archimedes' principle dictates that an object immersed in a fluid (in this case, air) experiences an upward buoyant force equal to the weight of the fluid displaced by the object. Helium, being significantly less dense than air, displaces a volume of air that weighs more than the helium itself. This difference in weight creates the upward buoyant force that allows helium balloons to float.
Key Factors Determining Lift:
Several factors interact to determine the precise amount of helium needed to lift a given weight:
-
Helium Density: The density of helium varies slightly with temperature and pressure. Colder temperatures lead to denser helium, resulting in slightly less lift. Conversely, warmer temperatures reduce helium's density, increasing lift capacity.
-
Air Density: Similarly, the density of air fluctuates with temperature, pressure, and humidity. Higher air density means greater buoyant force for the same volume of helium. High altitude, where air density is lower, will require more helium to achieve the same lift.
-
Balloon Material: The weight of the balloon itself significantly impacts the overall lift. Lighter balloon materials, such as Mylar, provide a greater net lifting force compared to heavier rubber balloons. The balloon's size also plays a crucial role, as a larger balloon displaces more air.
-
Payload Weight: This includes not only the object you want to lift but also the weight of any strings, attachments, and the balloon itself.
-
Helium Purity: The purity of the helium gas directly affects its density. Impurities reduce the lifting capacity. Using high-purity helium is essential for precise lift calculations.
Calculating the Required Helium: A Step-by-Step Approach
Accurately calculating the amount of helium needed involves a few key steps and considerations. While a precise calculation requires specialized tools and knowledge, we can develop an approximation using readily available data.
Step 1: Determining the Lifting Force of Helium
First, we need to determine the lifting power of a given volume of helium. This varies based on the conditions mentioned above. However, a reasonable approximation under standard atmospheric conditions (sea level, 15°C, and standard atmospheric pressure) is that one cubic foot of helium can lift approximately 0.069 pounds. This is a simplified approximation, and the actual lifting force may vary.
This figure represents the difference in weight between the displaced air and the helium itself. The exact number can be refined using a more detailed calculation considering the precise temperature, pressure, and humidity.
Step 2: Accounting for Payload and Balloon Weight
Next, we must account for the total weight you need to lift. This includes:
- Target Weight: The one pound you intend to lift.
- Balloon Weight: The weight of the balloon material itself. This can vary significantly depending on size and material. Consult the manufacturer's specifications or weigh the balloon to get an accurate measurement.
- Attachment Weight: The weight of any strings, ropes, or other attachments used to secure the payload.
Step 3: Calculating the Required Helium Volume
To calculate the total volume of helium required, use the following formula:
Total Helium Volume (cubic feet) = (Target Weight + Balloon Weight + Attachment Weight) / Lifting Force per Cubic Foot of Helium
Using our approximation of 0.069 pounds of lifting force per cubic foot:
Total Helium Volume (cubic feet) ≈ (1 lb + Balloon Weight + Attachment Weight) / 0.069 lb/cu ft
Step 4: Converting Volume to Mass
Once you have the required helium volume in cubic feet, you can convert it to mass (typically measured in grams or kilograms). This conversion requires knowing the density of helium under the prevailing atmospheric conditions. The density of helium at standard atmospheric conditions is approximately 0.011 kg/m³.
Remember to use consistent units throughout your calculations (e.g., pounds, cubic feet, kilograms, cubic meters).
Practical Considerations and Safety Precautions
While the calculations provide a theoretical estimate, several practical considerations influence the actual amount of helium needed:
-
Helium Loss: Helium is a lightweight gas and can escape through small openings or pores in the balloon material. Allow for some helium loss, especially with time.
-
Environmental Factors: Temperature, pressure, and humidity significantly impact both air and helium density, affecting the lift capacity. Account for variations from standard conditions.
-
Balloon Inflation: Ensure the balloon is inflated correctly to its optimal capacity. Overinflation can cause the balloon to burst, and underinflation will reduce the lifting power.
-
Safety: Always handle helium with caution. It is an asphyxiant gas, and its accumulation in enclosed spaces can displace oxygen, causing breathing difficulties. Ensure adequate ventilation and never use helium in situations that may risk asphyxiation.
-
Legal Considerations: Always check local regulations regarding helium use, particularly in large-scale applications.
Advanced Calculations and Professional Assistance
For more accurate calculations, especially for heavier payloads or applications requiring a high degree of precision, you may need to consult specialized resources or seek professional assistance. Software packages and engineering handbooks often provide more sophisticated tools and databases for precise calculations of buoyancy under varied atmospheric conditions.
Remember, the 0.069 lb/cu ft value is a simplified approximation. Actual values may deviate due to changes in temperature, pressure, and humidity. Professional engineers, particularly in aerospace or balloon industries, possess the knowledge and resources to perform accurate buoyancy calculations for complex scenarios.
Conclusion
Determining the precise amount of helium to lift one pound requires careful consideration of several interacting factors. While the simplified calculations provide a reasonable estimate, practical considerations and potentially significant variations in atmospheric conditions require a cautious and informed approach. Always prioritize safety and consider seeking professional advice for projects involving significant weight or high accuracy requirements. This comprehensive guide provides a solid foundation for understanding the physics involved and conducting initial estimations. However, remember that practical experimentation and professional guidance are crucial for successful and safe applications.
Latest Posts
Latest Posts
-
500 Yards Is How Many Miles
Apr 05, 2025
-
What Grade Is 57 Out Of 75
Apr 05, 2025
-
37 Months Is How Many Years
Apr 05, 2025
-
How Many Days Since May 28th
Apr 05, 2025
-
Cubic Feet Per Gallon Of Water
Apr 05, 2025
Related Post
Thank you for visiting our website which covers about How Much Helium To Lift 1 Pound . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.
